Hard water may cause many problem. Therefore, it is important to have a simple method to determine hardness. Such method is based on a titration of the water sample with a solution of acomplexing agent, named ethylenediaminetetraacetate (EDTA). The common form of the agent is disodium salt Na2H2EDTA. It is colorless. It can be weighed and dissolve in water to form a stable solution. At high pH (> 10) the remaining protons leave EDTA forming EDTA4- anion:
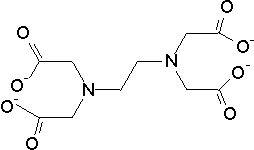
Mg2+ + EDTA4- ® MgEDTA2-
All reactants are colorless as well. Therefore, to detect the end point of this reaction we need an indicator. For this purpose we will use an organic dye named Eriochrome Black T . This compound forms pink complexes with Mg2+ ions and with Ca2+ ions. Aqueous solutions of the indicator at pH 10 are blue. Thus when pink complex MgInd reacts with EDTA, it results in the blue solution of indicator:
MgInd + EDTA4- ® MgEDTA2- + Ind (charges are not balanced!)
The reaction occurs because the EDTA forms much more stable complex than the indicator. A similar equation can be written for calcium.
Reagents:
Standard EDTA solution.
Buffer (pH 10) : mixture of ammonia and ammonium chloride solutions
in water.
Eriochrome Black T
Buret
Procedure
A 50 mL sample of tap water is usually reasonable. To the sample, add
5 mL of buffer, and 3-5 drops of indicator. Titrate with EDTA from a 50
mL buret and note when the color changes from red to blue. Repeat the titration
two more times to accurately measure the total Mg2+ and Ca2+
concentration.
Calculations:
At the equivalence point, number of moles of Mg before titrations is
exactly equal to the number of moles of EDTA you have added.
NMg = NEDTA
Number of moles of any compound in solution NX can be written
as
NX = CX VX, where CX is
molarity, and VX is volume. Thus,
CMgVMg = CEDTA VEDTA,
and CMg = CEDTA VEDTA/ VMg
Experiment 4
Name __________________________ Date _________________
Concentration of EDTA
Volume of Mg sample
Volume of EDTA
Titration No. 1:
Titration No. 2
Titration No. 3:
Average
Concentration of Mg (mMoles/L):